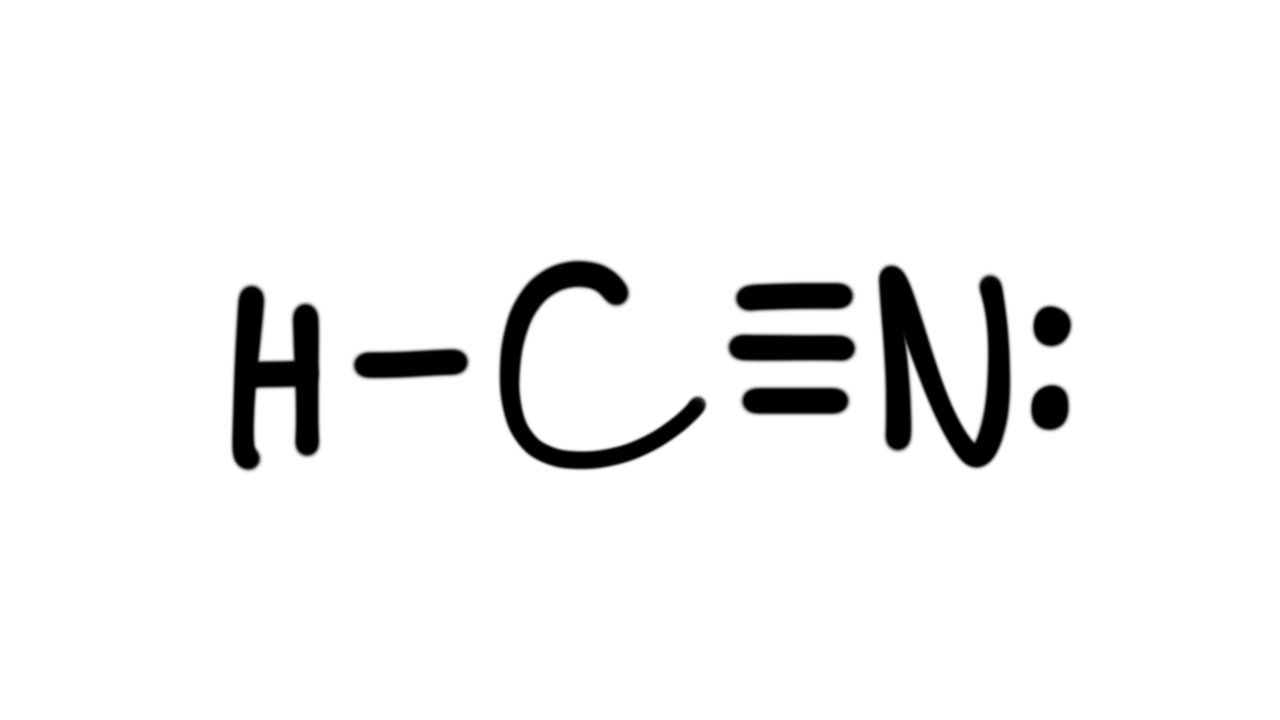Draw The Lewis Structure For Hcn
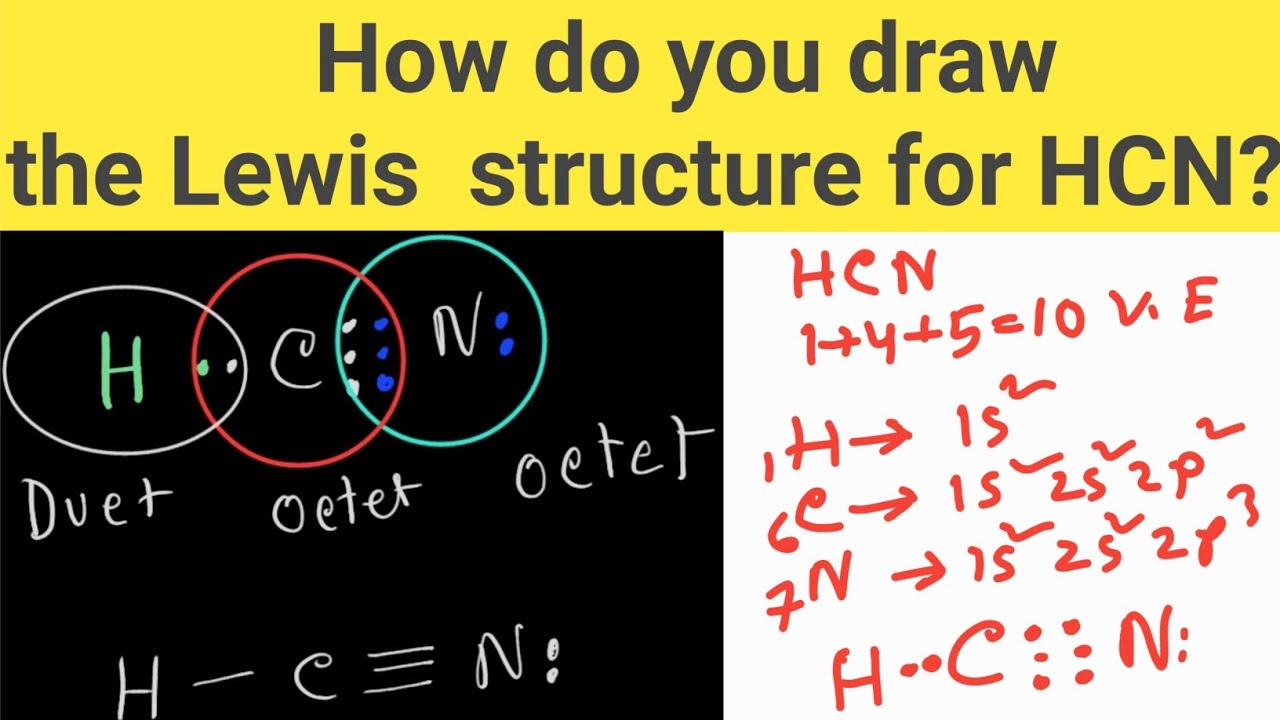
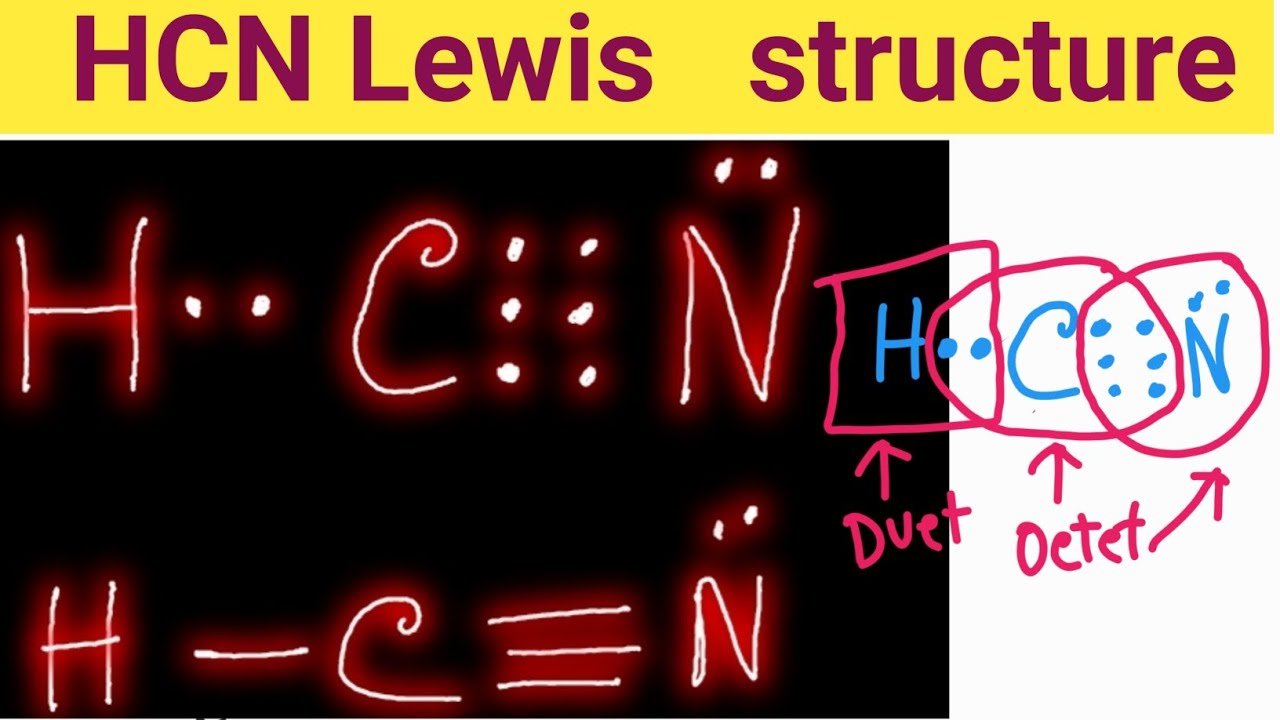
Draw The Lewis Structure For Hcn - Add the valence electrons of all the molecules' atoms: Count the valence electrons you can. Put one electron pair in each bond4. Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon, and one nitrogen atom. Put least electronegative atom in centre3. • how to draw lewis structure for pocl3. We'll also compare hnc to hcn and discuss why both are of. Does this compound have any plausible. Follow the steps to mark lone pairs,. Ch 4 has 4 valence electrons in c, and 1. Ch 4 has 4 valence electrons in c, and 1. Web draw the most likely structure for hcn based on formal charges, showing the formal charge on each atom in your structure. Web in this video, we will look at the lewis structure of hydrogen cyanide having a chemical formula of hcn. Count the valence electrons you can. Web with the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. Web draw out a correct lewis structure for the following compounds. Put least electronegative atom in centre3. We'll also compare hnc to hcn and discuss why both are of. Put the least electronegative atom c in the middle with h and cl on either side. Web how to draw a lewis structure for hcn? Web we show two methods to find correct lewis structure of hcn. Web learn about the lewis structure of hcn, including its bonding properties, electron configuration, and frequently asked questions. One uses math, the other puzzle pieces to give the three correct structure. We'll also compare hnc to hcn and discuss why both are of. Does this compound have any. • how to draw lewis structure for pocl3. Ch 4 has 4 valence electrons in c, and 1. Describe the interactions between atoms using lewis structures (what happens to the valence electrons) Web draw out a correct lewis structure for the following compounds. Web the lewis structure (lewis dot diagram) for hcn.1. / @drmasi thank you for watching dr. Describe the interactions between atoms using lewis structures (what happens to the valence electrons) Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon, and one nitrogen atom. Web draw the most likely structure for hcn based on formal charges, showing the formal charge on each. We'll also compare hnc to hcn and discuss why both are of. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Web in this example problem, we draw the lewis structure for hcn, which has a triple bond. Web for the hcn lewis structure,. Add the valence electrons of all the molecules' atoms: / @drmasi thank you for watching dr. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Note that hydrogen is often. Web the lewis structure (lewis dot diagram) for hcn.1. / @drmasi thank you for watching dr. Web in this example problem, we draw the lewis structure for hcn, which has a triple bond. Web draw the lewis structures of ch 4, pcl 3, co 2, and hcn. Put least electronegative atom in centre3. Add the valence electrons of all the molecules' atoms: Put one electron pair in each bond4. There is also a video and a study. Web draw a skeleton structure. Web we show two methods to find correct lewis structure of hcn. Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon, and one nitrogen atom. Web establish a general procedure for drawing lewis structures; Put the least electronegative atom c in the middle with h and cl on either side. Web draw out a correct lewis structure for the following compounds. Follow the steps to mark lone pairs,. Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon,. Web the lewis structure (lewis dot diagram) for hcn.1. The molecule is made up of one hydrogen atom,. Web we show two methods to find correct lewis structure of hcn. Web in this example problem, we draw the lewis structure for hcn, which has a triple bond. There is also a video and a study. Describe the interactions between atoms using lewis structures (what happens to the valence electrons) Follow the steps to mark lone pairs,. Note that hydrogen is often. Web how to draw a lewis structure for hcn? Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon, and one nitrogen atom. There is also a video and a study. Note that hydrogen is often. Follow the steps to mark lone pairs,. Put the least electronegative atom c in the middle with h and cl on either side. Web we show two methods to find correct lewis structure of hcn. Put one electron pair in each bond4. • how to draw lewis structure for pocl3. Does this compound have any plausible. Put least electronegative atom in centre3. We'll also compare hnc to hcn and discuss why both are of. Web learn about the lewis structure of hcn, including its bonding properties, electron configuration, and frequently asked questions. Web to draw the lewis structure of an atom, write the symbol of the atom and draw dots around it to represent the valence electrons. Web in this video, we will look at the lewis structure of hydrogen cyanide having a chemical formula of hcn. Web with the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. Web learn how to draw the lewis structure for hcn, a molecule with one hydrogen, one carbon, and one nitrogen atom. Describe the interactions between atoms using lewis structures (what happens to the valence electrons)37. Lewis Dot Structure of HCN How to Draw Lewis Structures Class 11
CHEMISTRY 101 Drawing Lewis Structures Multiple bonds, HCN YouTube
How do you draw the Lewis structure of HCN (hydrogen cyanide)? HCN
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw
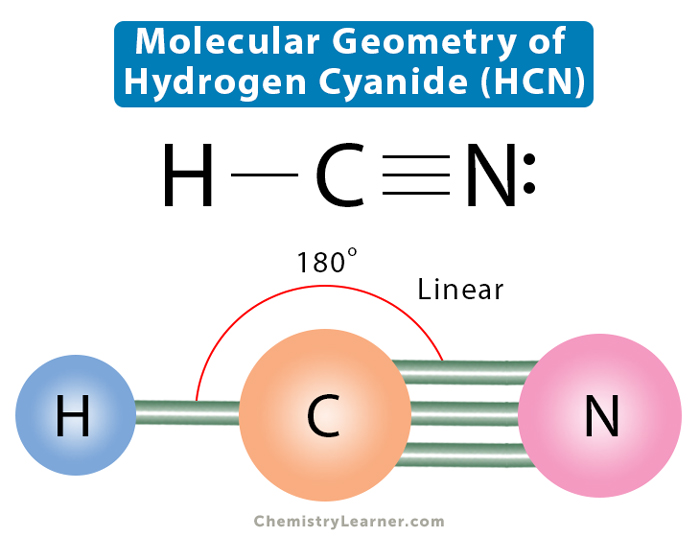
HCN Lewis Structure Molecular Geometry
Lewis Diagram For Hcn
Lewis Diagram For Hcn Draw Easy
HCN Lewis Structure, Molecular Geometry, Hybridization, MO Diagram, and
Draw The Lewis Dot Structure Of Hydrogen Cyanide Hcn vrogue.co
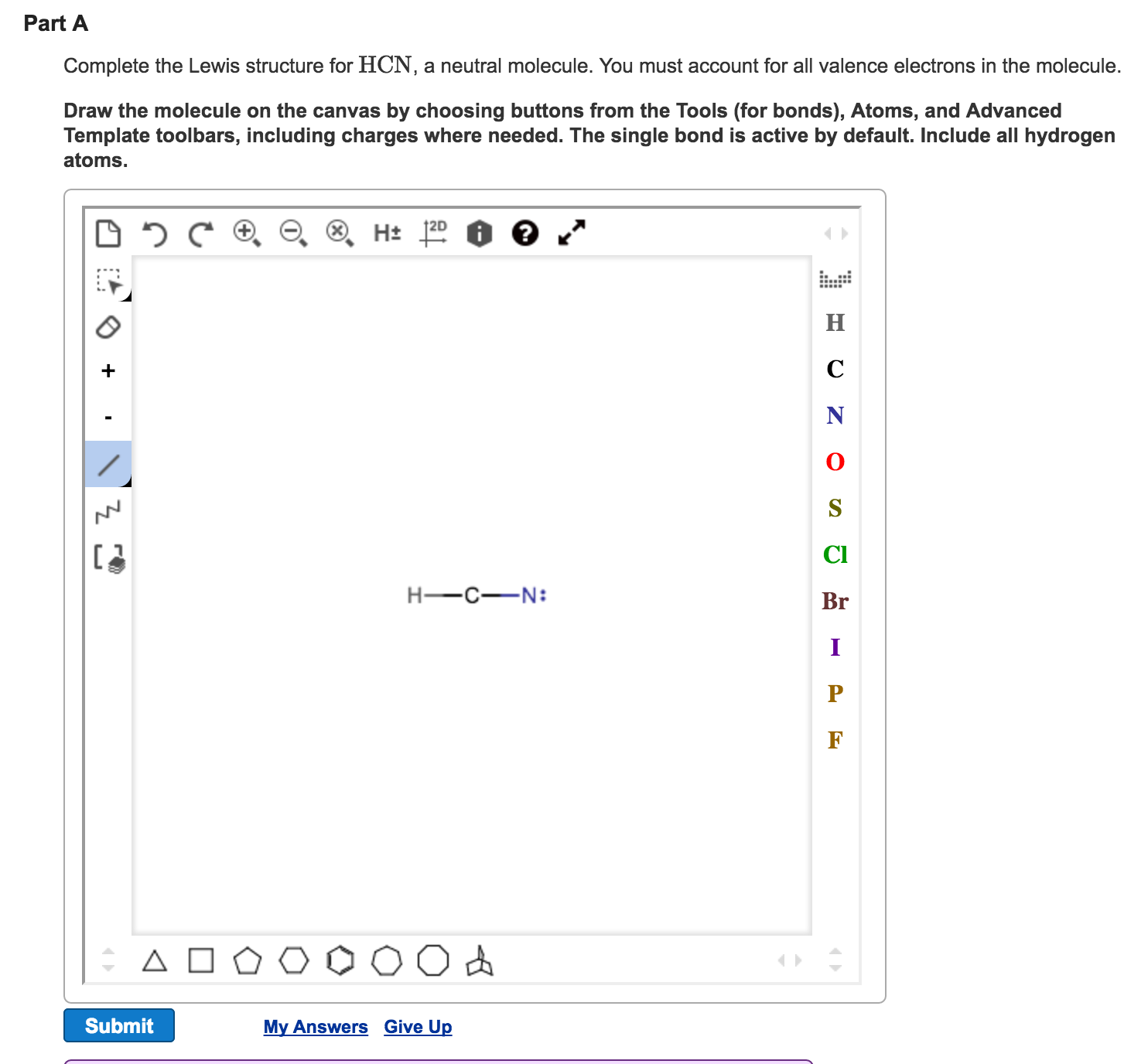
Solved Complete the Lewis structure for HCN, a neutral
Web For The Hcn Lewis Structure, Calculate The Total Number Of Valence Electrons For The Hcn Molecule.
Ch 4 Has 4 Valence Electrons In C, And 1.
Web Draw Out A Correct Lewis Structure For The Following Compounds.
Web In This Example Problem, We Draw The Lewis Structure For Hcn, Which Has A Triple Bond.
Related Post: